카이스트, 꿈의 배터리 개발 성공
기사입력 2022.01.14 15:31
▲개발된 엘라스토머 기반 고체전해질을 이용해 구현된 전고체전기 및 기대효과(그림 출처 : 카이스트)
전기차 주행거리 확보·안전 문제 해결
이차전지 발전에 새로운 가능성 제시
카이스트와 조지아공대 연구팀이 한 번의 충전으로 서울·부산을 왕복할 수 있도록 하는 세계 최고성능 전고체전지를 개발해 ‘꿈의 배터리’ 기술을 실현시켰다.
카이스트 생명화학공학과 김범준 교수 연구팀이 미국 조지아공대 이승우 교수팀과 공동연구를 통해 새로운 개념의 엘라스토머 고분자 전해질을 개발하고 이를 통해 세계 최고성능의 전고체전지를 구현했다고 13일 밝혔다.
전고체 리튬메탈전지(all-solid-state Li-metal battery)는 이차전지에 사용되는 휘발성이 높은 액체전해질을 고체로 대체해 화재 및 자동차 안전사고를 막을 수 있는 미래기술이다.
현재 상용화된 리튬이온전지(Li-ion battery)에 비해 에너지밀도를 획기적으로 향상해 자동차 주행거리 확보 및 안전 문제를 해결할 수 있는 ‘꿈의 배터리 기술’이다.
공동 연구팀은 상온에서 리튬(Li) 이온의 전도도가 탁월하며, 기계적 신축성이 모두 확보된 엘라스토머(고무) 형태의 고분자 전해질을 개발했다.
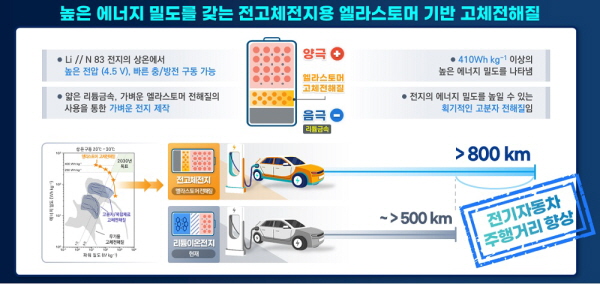
이를 전고체전지에 적용해 410Wh/kg의 세계 최고성능을 보이는 전고체 리튬 메탈전지를 구현했다.
이러한 기술을 도입하면 현재 한번 충전으로 800km까지 주행 가능한 전기자동차의 구현(현재 500 km수준)이 가능할 것으로 보이며, 기존의 액체 전해질을 적용한 리튬이온전지의 안정성을 획기적으로 향상할 것으로 기대된다.
연구팀은 고무처럼 신축성이 탁월한 엘라스토머 내부에 리튬 이온전도도가 매우 높은 플라스틱 결정 물질을 3차원적으로 연결한 엘라스토머 고분자 고체전해질을 개발했다.
연구팀이 개발한 전해질은 기존에 대표적인 폴리에틸렌옥사이드(PEO) 기반의 고분자 전해질에 비해 100배 정도 향상된 10-3 S/cm의 이온전도도를 가진다.
또 고무처럼 신축성이 우수한 전해질은 전지 충‧방전 시 안정성에 가장 큰 문제가 되는 리튬 덴드라이트의 성장을 억제해 탁월한 전지 성능 및 안정성을 확보했다.
개발된 고분자 전해질은 얇은 리튬금속 음극과 니켈 리치 양극(NCM-Ni83)으로 구성된 전고체전지에서 4.5V 이상의 고전압에서도 안정적인 구동을 보였고, 410Wh/kg 이상의 세계 최고의 에너지밀도를 보였다.
이차전지 분야 권위자인 강기석 서울대 교수는 “전고체 이차전지에 대한 세계적인 개발 경쟁이 치열하다”며 “연구팀의 이번 개발은 이차전지 발전에 새로운 가능성을 제시할 것”이라고 말했다.
김범준 교수는 "엘라스토머 전해질이라는 기존과는 완전히 다른 새로운 종류의 고체전해질을 개발해 소재 원천 기술을 확보했다는 것에 큰 의의가 있다ˮ라고 밝혔다.
한편 카이스트 한정훈 및 조지아공대 이승훈 연구원이 공동 제1 저자로 참여한 이번 연구는 국제 학술지 `네이처(Nature)' 1월 13일에 출판됐다.
이번 연구에는 한국연구재단의 중견도약연구사업, 미래소재디스커버리 사업과 기초연구실지원사업의 지원을 받아 수행되었으며, 한국화학연구원의 김병각 박사, 한국에너지기술연구원의 정규남 박사가 공동연구에 참여했다.
카이스트 생명화학공학과 김범준 교수 연구팀이 미국 조지아공대 이승우 교수팀과 공동연구를 통해 새로운 개념의 엘라스토머 고분자 전해질을 개발하고 이를 통해 세계 최고성능의 전고체전지를 구현했다고 13일 밝혔다.
전고체 리튬메탈전지(all-solid-state Li-metal battery)는 이차전지에 사용되는 휘발성이 높은 액체전해질을 고체로 대체해 화재 및 자동차 안전사고를 막을 수 있는 미래기술이다.
현재 상용화된 리튬이온전지(Li-ion battery)에 비해 에너지밀도를 획기적으로 향상해 자동차 주행거리 확보 및 안전 문제를 해결할 수 있는 ‘꿈의 배터리 기술’이다.
공동 연구팀은 상온에서 리튬(Li) 이온의 전도도가 탁월하며, 기계적 신축성이 모두 확보된 엘라스토머(고무) 형태의 고분자 전해질을 개발했다.
이를 전고체전지에 적용해 410Wh/kg의 세계 최고성능을 보이는 전고체 리튬 메탈전지를 구현했다.
이러한 기술을 도입하면 현재 한번 충전으로 800km까지 주행 가능한 전기자동차의 구현(현재 500 km수준)이 가능할 것으로 보이며, 기존의 액체 전해질을 적용한 리튬이온전지의 안정성을 획기적으로 향상할 것으로 기대된다.
연구팀은 고무처럼 신축성이 탁월한 엘라스토머 내부에 리튬 이온전도도가 매우 높은 플라스틱 결정 물질을 3차원적으로 연결한 엘라스토머 고분자 고체전해질을 개발했다.
연구팀이 개발한 전해질은 기존에 대표적인 폴리에틸렌옥사이드(PEO) 기반의 고분자 전해질에 비해 100배 정도 향상된 10-3 S/cm의 이온전도도를 가진다.
또 고무처럼 신축성이 우수한 전해질은 전지 충‧방전 시 안정성에 가장 큰 문제가 되는 리튬 덴드라이트의 성장을 억제해 탁월한 전지 성능 및 안정성을 확보했다.
개발된 고분자 전해질은 얇은 리튬금속 음극과 니켈 리치 양극(NCM-Ni83)으로 구성된 전고체전지에서 4.5V 이상의 고전압에서도 안정적인 구동을 보였고, 410Wh/kg 이상의 세계 최고의 에너지밀도를 보였다.
이차전지 분야 권위자인 강기석 서울대 교수는 “전고체 이차전지에 대한 세계적인 개발 경쟁이 치열하다”며 “연구팀의 이번 개발은 이차전지 발전에 새로운 가능성을 제시할 것”이라고 말했다.
김범준 교수는 "엘라스토머 전해질이라는 기존과는 완전히 다른 새로운 종류의 고체전해질을 개발해 소재 원천 기술을 확보했다는 것에 큰 의의가 있다ˮ라고 밝혔다.
한편 카이스트 한정훈 및 조지아공대 이승훈 연구원이 공동 제1 저자로 참여한 이번 연구는 국제 학술지 `네이처(Nature)' 1월 13일에 출판됐다.
이번 연구에는 한국연구재단의 중견도약연구사업, 미래소재디스커버리 사업과 기초연구실지원사업의 지원을 받아 수행되었으며, 한국화학연구원의 김병각 박사, 한국에너지기술연구원의 정규남 박사가 공동연구에 참여했다.
많이 본 뉴스
[열린보도원칙] 당 매체는 독자와 취재원 등 뉴스이용자의 권리 보장을 위해 반론이나 정정보도, 추후보도를 요청할 수 있는 창구를 열어두고 있음을 알려드립니다.
고충처리인 장은성 070-4699-5321 , news@e4ds.com


.jpg)