전기연, 비싼 바나듐 대체 아연-망간 레독스전지 개발
기사입력 2023.01.10 10:31
최적 비율 촉매, 전지 성능 높이고 가격 경쟁력 확보
한국전기연구원(KERI, 원장 직무대행 김남균)이 고가의 바나듐을 대체해 아연과 망간을 최적 비율로 이용해 전지 성능을 높이고 가격 경쟁력을 확보해 향후 화재 및 폭발 없는 친환경 전지가 대중화 될 것으로 기대된다.
KERI는 차세대전지연구센터 박준우 박사팀과 부산대 박민준 교수팀이 기존 레독스흐름전지의 한계를 뛰어넘는 기술을 개발했고, 관련 논문은 우수한 평가를 받아 국제 저명 학술지에 게재됐다고 9일 밝혔다.
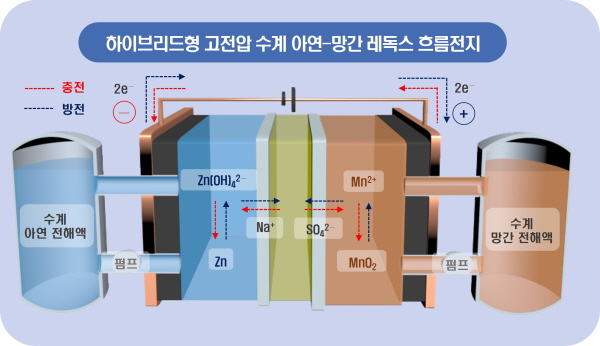
레독스흐름전지(Redox Flow Battery)는 환원(Reduction), 산화(Oxidation), 흐름(Flow)의 단어를 합성한 용어로써, 산화·환원이라는 화학적 반응을 통해 전자가 전해액의 도움을 받아 음극(-)에서 양극(+)으로 이동하며 전기 에너지를 발생시키는 원리다.
기존 이차전지와 달리 화학적 반응이 일어나는 부분과 전기를 저장하는 부분을 구분했기 때문에 출력과 용량의 독립적인 설계가 가능하고, 전지의 대용량화가 가능하다.
또한 배기가스를 배출하지 않고, 화재·폭발 위험도 없다.
현재 레독스흐름전지의 주요 핵심소재인 ‘바나듐’은 우리가 중국 등 수입에 의존하는 고(高)가격의 금속이다. 전지의 성능을 좌우하는 전압도 바나듐 레독스흐름전지가 상용 리튬이차전지보다 낮아 효율성이 10∼15% 정도 떨어진다.
이에 KERI-부산대가 활용한 대체 금속은 상대적으로 저렴하고, 수급도 안정적인 망간과 아연이다.
특히 하이브리드형 레독스흐름전지는 독특한 이중 이온교환막 구조로 구성돼 알칼리성의 아연 전해액과 산성의 망간 전해액의 동시 사용이 가능하고, 높은 전위차를 기대할 수 있다.
물론 어려움도 있었다. 망간 양극 활물질이 산화되었을 때 원래대로 환원되지 않고, 전극 및 이온 교환막 표면에 증착(deposition)되는 문제가 발생한 것이다.
연구팀은 망간의 낮은 가역성(reversibility) 문제를 개선하기 위해 ‘탄소 환원법’을 활용해 비스무트(Bismuth) 금속을 전극 표면에 증착시켰고, 니켈과 마그네슘, 구리, 코발트 이온 등이 첨가된 전해액을 활용했다.
비스무트는 전기 전도성이 우수해 전자 이동을 향상시켰고, 전기화학 성능 특성 분석을 통해 최적의 비율로 만들어진 전해액은 촉매 역할을 수행하여 전지의 안정성(활물질 부반응 억제)과 성능, 수명을 크게 높였다.
이후 투과전자현미경(Transmission Electron Microscopy), X선광전자분광법(X-ray Photoelectron Spectroscopy), 플라즈마 발광(ICP-OES) 등 다양한 분석으로 개발 기술의 효과가 검증됐다.
그 결과 KERI-부산대의 하이브리드형 아연-망간 레독스흐름전지는 기존 바나듐계보다 높은 전압(1.3V -> 2.52V) 구현, 에너지 효율 10% 이상 향상 등의 결과를 확보할 수 있었다.
KERI 박준우 박사는 “저비용의 아연/망간 소재와 가역성을 높일 수 있는 금속 이온 촉매 기술이 결합된 신개념의 레독스흐름전지 기술”이라며 “이번 성과는 차세대 장주기 대용량 레독스흐름전지의 보급 확대에 크게 기여할 것”이라고 전했다.
관련 연구결과는 높은 우수성을 인정받아 에너지 분야 국제 전문 학술지인 에너지 스토리지 머티리얼스(Energy Storage Materials)에 최근 논문이 게재(Impact Factor 20.831/JCR 4.2%)됐다.
KERI는 지난해 구축한 국내최초 ‘광주 레독스흐름전지 시험인증센터’를 활용해 이번 개발 기술을 더욱 확장·발전시킬 예정이며, 상용화의 관건인 실제 대면적/고농도 셀 제작까지 이어질 수 있도록 만든다는 목표다. 또한, 관련 수요업체도 발굴하여 기술이전을 추진할 계획이다.
한편 KERI는 과학기술정보통신부 국가과학기술연구회 산하 정부출연연구기관이다. 이번 연구는 산업부 지역산업거점기관지원사업, 한국연구재단 우수신진연구사업 및 기초연구실사업으로 수행됐다.
많이 본 뉴스
[열린보도원칙] 당 매체는 독자와 취재원 등 뉴스이용자의 권리 보장을 위해 반론이나 정정보도, 추후보도를 요청할 수 있는 창구를 열어두고 있음을 알려드립니다.
고충처리인 장은성 070-4699-5321 , news@e4ds.com




.jpg)

