[양자 톺아보기] 2. 모든 것은 원자로 이루어졌다
기사입력 2018.08.16 08:50
인공지능, 가상현실 등의 발달로 처리할 데이터양은 늘어가는데 집적회로의 한계는 가까워지고 있다. 그래서 트랜지스터로 만들어진 게이트 대신 양자를 연산법칙으로 사용하는 양자 컴퓨터가 대안으로 떠오르고 있다. 도대체 양자가 뭔지, 또 그걸로 어떻게 하기에 대안이라는 걸까? 과학과 인연이 없던 기자가 양자부터 최근 이슈가 되고 있는 양자 컴퓨터까지, 배우는 마음으로 차근차근 들여다본다.
지구 문명이 모조리 파괴되었을 때, 후세를 위해서 딱 한 마디만 남길 수 있다면 무슨 말을 남기겠냐는 질문에 물리학자 리처드 파인만은 이렇게 답했다. "모든 것은 원자로 이루어져 있다!" 그 말대로다. 우리를 포함한 우리 주변의 모든 것은 원자로 이루어져 있다. 그리고 양자역학은 그 원자의 움직임을 다루는 학문이다. 그렇다면 양자역학을 알기 위해선 원자에 대해서 알아보는 게 순서일 것이다.
원자, 가장 작은 존재
고대 그리스의 철학자 데모크리토스는 기원전 5세기경, 모든 물질이 더 이상 쪼갤 수 없는 작은 것들로 이루어졌다는 원자설을 주장했다. 하지만 당시로선 실증할 수 없었기 때문에 철학자들 사이에서 철학적인 논의만 오갔다. 이후 중세를 거치면서 원자설은 수면 아래로 가라앉았다.
원자설을 다시 수면 위로 끌어올린 것은 영국의 화학자이며 물리학자인 존 돌턴이다. 돌턴은 1803년, 자신의 저서 ‘화학의 신체계’에서 ‘질량 보존의 법칙’과 ‘일정 성분비의 법칙’이 성립하는 이유를 설명하기 위해 원자설을 사용했다. 그의 원자설에 따르면, 원자는 성질을 갖고 있는 원소의 가장 작은 기본 입자로 더 이상 쪼갤 수 없는 것이었다. 당시 많은 과학자들은 돌턴의 원자설을 유용한 가설로 사용하였지만, 원자가 실존한다고는 생각하지 않았다. 이 추세에 반기를 든 것이 바로 독일의 루트비히 볼츠만이다.
인정받은 원자
볼츠만은 19세기 말, 원자의 존재를 확신하고 원자설이 아닌 원자론에 기반을 둔 기체분자운동론을 주장했다. 또한 원자와 분자를 통계적으로 다루는 통계역학의 기초를 닦았다. 그러나 물질이 공간상에 불연속적으로 존재한다는 믿음으로부터 출발한 볼츠만의 주장은 물질이 공간상에 연속적으로 존재한다는 경험적인 믿음을 가지고 있던 과학자들로부터 통렬한 비판을 받았다. 특히 경험적으로 측정할 수 있는 것만 과학이라 주장한 물리학자 겸 철학자, 에른스트 마흐는 볼츠만의 면전에서 대놓고 ‘나는 원자론을 믿지 않는다.’라고 말할 정도였다. 볼츠만은 시류에 반박하기 위해 철학마저 연구했으나 1906년, 우울증으로 자살하고 만다.

루트비히 볼츠만 "왜 나만..."
공교롭게도 볼츠만이 자살하기 1년 전, 볼츠만의 주장을 뒷받침해줄 논문이 발표되었다. 스위스 베른에서 물리학을 전공한 특허청 심사관이 유체 안에 존재하는 입자가 불규칙적으로 움직이는 현상인 ‘브라운 운동’을 설명하기 위해선 원자가 반드시 존재해야 한다는 것을 입증한 것이다. 그 심사관이 바로 아인슈타인이었다. 이후 프랑스의 장 바티스트 페렝이 이를 실험으로 검증해내면서 원자의 존재는 경험적으로 증명되었다.
원자보다 더 작은 것들
몇몇 과학자들이 원자의 존재를 부인하고 있을 때, 원자를 연구하던 과학자들은 더 작은 것들을 찾아냈다. 조지프 존 톰슨은 1897년, 음극선을 연구하다 전자를 발견했다. 보통의 기체나 액체는 전기를 띠고 있지 않다. 즉 전기적으로 원자는 중성이다. 그런데 톰슨이 발견한 전자는 음전기를 띄고 있었다. 그렇다면 원자의 음전기를 없애는 양전기가 원자 어딘가 있지 않을까? 1906년, 톰슨은 양전하를 가지는 물질 속에 양전하의 전체 전하량과 동일한 전하량만큼의 전자가 균일하게 분포하고 있는 원자 모형을 제시했다.
톰슨의 제자인 어니스트 러더퍼드는 원자의 내부 구조를 조사하기 위해 1909년부터 금박실험을 시작했다. 1/20000cm의 금박을 만들고 방사성 원소에서 나오는 알파입자(양성자 2개와 중성자 2개로 구성된 안정된 입자)를 초속 약 16,000km의 속도로 금박을 향해 발사했다. 발사 장치를 따로 만든 건 아니다. 그저 방사성 원소를 담아둔 통을 금박을 향해 열어 둔 것이다. 아무튼 러더퍼드는 톰슨의 원자 모형이 맞다면, 알파 입자를 금박을 향해 쏘는 것은 탁구공을 여러 겹 쌓은 곳에 야구공을 던지는 것과 같다고 생각했다. 거의 대부분의 알파입자는 예상대로 금박을 그대로 통과했다. 하지만 몇몇 알파입자는 큰 각도로 튕겨 나갔고 어떤 알파입자는 아예 뒤쪽으로 튕겨 나갔다.
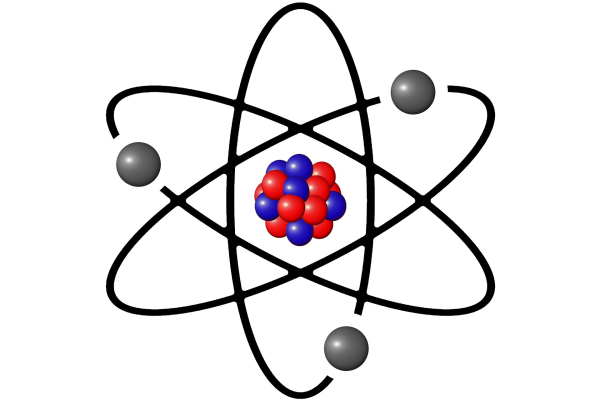
러더퍼드의 원자 모형
러더퍼드는 원자 내부는 거의 비어 있지만, 매우 작고 단단한 무언가 존재해 알파 입자를 튕겨낸다는 사실을 알아냈다. 러더퍼드는 단단한 무언가를 원자핵이라 명명한 다음 새로운 원자 모형을 만들었다. 우리가 익히 아는 행성 궤도를 닮은 원자 모형이다. 러더퍼드의 원자 모형에 따르면 전자가 원자핵을 맴돌지만 서로의 간격은 굉장히 멀다. 원자가 돌과 같다던 돌턴의 추측과 건포도 붙은 빵과 같다던 톰슨의 원자 모형은 완전히 폐기된 것이다.
원자가 무너지지 않아
앞서 살펴봤듯이 원자는 원자핵과 전자로 이루어져 있다. 원자핵은 원자의 10만분의 1 크기이며, 전자는 원자핵의 1000분의 1 크기다. 보통 전자의 궤도를 원자의 크기로 본다. 원자를 1000000000000000배 확대했을 때 원자핵과 전자는 100km만큼 떨어져 있다. 그리고 나머지 공간은 전부 비어 있다. 따라서 이런 의문이 생길 수 있다.
앤트맨이 작아지는 장면 "핌 입자, 핌 입자가 필요해!"
원자 내의 빈 공간이 저리 넓은 데 어째서 원자는 쪼그라들지 않을까? 또 원자로 이루어진 만물은 어째서 서로를 통과하지 않고 부딪히는 것인가?
그 답을 알기 위해선 먼저 닐스 보어가 1913년에 제시한 원자 모형부터 톺아봐야 한다. 보어는 수소 원자의 불연속적인 스펙트럼을 관찰하고 그것을 토대로 모형을 만들었다. 전자는 일정한 궤도에서 원운동을 하며, 각 궤도는 연속적이지 않고 띄엄띄엄 떨어져 존재한다. 그러나 정해진 궤도가 있어도 원자끼리 부딪히지 않을 확률은 여전히 높다.
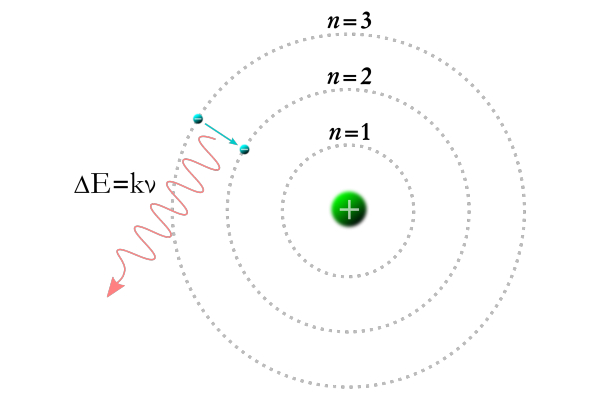
보어의 원자 모형
베르너 하이젠베르크는 여기서 궤도를 포기한다. 전자의 위치는 불확정적이어서 언제든지 여러 곳에서 동시에 나타날 수 있다. 직접 관측한다면 전자가 어디에 있는지 알 수 있지만, 관측하지 않는다면 여러 곳에 존재할 수 있다. 전자는 구름처럼 이리저리 흩어져 있다. 전자가 궤도만 돌 때보다 원자끼리 부딪힐 가능성이 더 높아졌다. 그러나 하이젠베르크의 이론대로라면 원자가 서로 부딪히더라도 튕겨 나간다고는 장담할 수 없다. 구름은 딱딱하지 않기 때문이다.
볼프강 파울리는 배타 원리로 원자를 딱딱하게 만들었다. 전자는 이미 자리를 차지한 전자의 자리를 뺏을 수 없다. 예를 들어 수소 원자핵 주변에는 하나의 전자만 존재한다. 다른 전자는 들어갈 수 없다. 한 점의 구름에는 한 개의 전자만 존재하며 두 개의 전자는 존재할 수 없다. 전자는 전자를 싫어한다. 원자끼리 부딪힐 수 밖에 없는 것이다. 하지만 원자핵은 꼭 전자와 짝을 이루려고 한다. 그래서 전자가 원자핵을 떠나지 않는다. 원자가 갑자기 허물어지거나 폭발하지 않는 이유다.
원자에 대해 대략 알아봤다. 이어질 기사에선 양자역학이 어떻게 시작되었는지에 대해 다뤄보겠다.
참고서적: "세상에서 가장 쉬운 양자역학 수업, 리먀오(고보혜), 더숲"
이수민 기자
- 1 Comments
- 주*규 (2018-08-14 오후 1:46:44)
- 그렇군요.
관련 웨비나
많이 본 뉴스
[열린보도원칙] 당 매체는 독자와 취재원 등 뉴스이용자의 권리 보장을 위해 반론이나 정정보도, 추후보도를 요청할 수 있는 창구를 열어두고 있음을 알려드립니다.
고충처리인 장은성 070-4699-5321 , news@e4ds.com

.JPG)


.png)
.jpg)






