UNIST, 암모니아 수소 생산기술 성공
기사입력 2021.05.07 13:54

▲(우측 하단부터 시계방향)양예진 연구원, 김정원 연구원, 조효이 연구원, 성아림 연구원, 김건태 교수
수전해比 전력량 3배 적어, 그린수소 시대 눈앞
수소 1.5배 수송·기존 액화암모니아 인프라 활용
국내 연구진이 액상 암모니아를 수소로 바꾸는 효율적인 기술을 개발하고, 최적의 공정 환경 등을 찾아낼 수 있는 새로운 분석기술까지 제시해 그린수소 시대를 앞당길 핵심 기술로 주목받고 있다.
UNIST(총장 이용훈) 에너지화학공학과 김건태 교수팀은 액상 암모니아(NH₃)를 전기로 분해해 순도 100%에 가까운 그린수소(H2)를 대량 생산하는 데 성공했다.
연구진이 제안한 분석기술로 평가한 결과 이 방식은 물 전기분해로 수소를 만드는 것 보다 소모 전력량이 3배나 적었다.
수소를 운송하는 방법 가운데 가장 효율적인 방법으로 암모니아 합성법이 있다. 수소로 암모니아를 합성하면 1.5배 많은 수소를 운반할 수 있고 운송 중 손실이 적기 때문이다.
또한 기존 액화 암모니아 운송 인프라를 활용할 수 있는 것도 장점이다.
반면에 수소를 암모니아로 합성하는 기술은 상용화되지만, 암모니아에서 다시 수소 뽑는 기술은 아직 초기 단계다.
연구팀은 다공성 니켈 폼(Ni foam) 전극을 이용해 액상 암모니아에서 수소를 추출하는 데 성공했다.
니켈 폼 전극 표면에서는 순환전류법(Cyclic Voltammetry)으로 고르게 도포된 촉매(백금) 입자가 있어 효율이 높다.
이렇게 합성된 전극을 액상 암모니아 넣은 뒤 전류를 흘려주면 액상 암모니아가 분해(전기분해)되면서 수소를 얻을 수 있다.
수소 순도를 나타내는 지표인 패러데이 효율은 90% 이상이었으며, 화학반응 속도를 나타내는 전류 밀도도 500mA cm-2 이상으로 높았다.
또한 가스 크로마토그래피(Gas chromatography, 가스 분석기)를 활용해 실시간으로 발생하는 가스양을 정량적으로 분석하는 프로토콜을 새롭게 제시해 효율적인 암모니아 전해 공정 조건(전해액의 산성도 등)도 찾아냈다.
최적화된 구동 환경에서는 1kWh의 전력으로 569L의 수소를 생산할 수 있다. 물 전기분해 대비 소비 전력량 3배 이상 낮은 수치다. 이는 암모니아를 쓸 때 물보다 적은 전력과 비용으로 그린수소를 생산할 수 있음을 실증한 것이다.
제 1저자인 양예진 UNIST 에너지공학과 석·박사통합과정 연구원은 “암모니아 전해 환경에서 뛰어난 수소 생산 활성도를 갖는 전극 제작 공법을 썼다”며 “이 전극은 산화 및 환원 반응에서 모두 함께 쓸 수 있는 전극으로 암모니아 전해 시스템 단순화 및 구축비용 절감에 큰 기여를 할 것으로 기대한다”고 말했다.
공동 제1저자 김정원 UNIST 에너지공학과 석·박사통합과정 연구원은 “이번 연구는 암모니아 전해 구동을 위한 최적의 조건을 찾은 것에도 의미가 크다”며 “향후 암모니아 전해 상용화를 위한 기초 연구로써도 의미가 있다”고 설명했다.
김건태 교수는 “이번 연구에서 제시한 고효율 전극을 암모니아 전해 공정을 적용하게 된다면 암모니아 전해 수소 생산 기술 상용화가 한층 빨라질 것”이라며 “암모니아, 더 나아가 폐(廢) 암모니아를 ‘CO2-free 수소’로 재 자원화하는 연구 가능성도 다각적으로 제시돼 신재생에너지의 새로운 패러다임 개척했다”고 전했다.
이번 연구는 롯데케미칼 기초소재연구소 이민재, 서명기, 민형기, 최영헌 박사가 각각 공동 저자로 참여했다. 연구결과는 에너지 및 재료 분야 국제학술지 재료화학 저널 A(Journal of Materials Chemistry A) 온라인판에 3월27일자로 공개됐으며 표지논문(inside front cover)으로 선정돼 정식출판을 앞두고 있다. 연구 수행은 롯데케미칼, 과학기술정보통신부-한국연구재단(NRF)등의 지원으로 이뤄졌다.
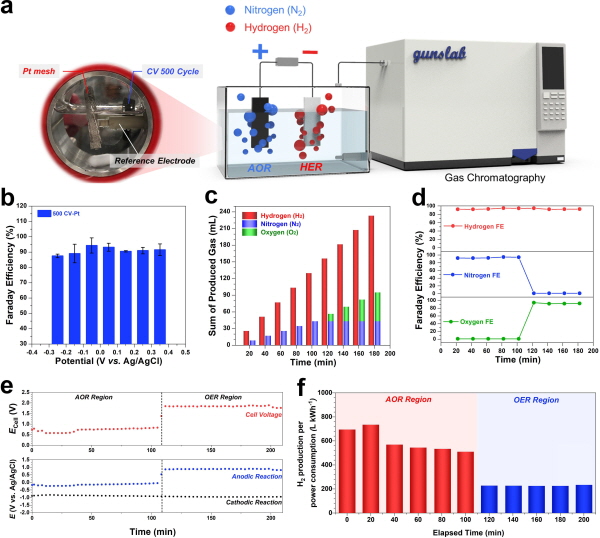
▲In-operando 암모니아 전해 공정 분석법
많이 본 뉴스
[열린보도원칙] 당 매체는 독자와 취재원 등 뉴스이용자의 권리 보장을 위해 반론이나 정정보도, 추후보도를 요청할 수 있는 창구를 열어두고 있음을 알려드립니다.
고충처리인 장은성 070-4699-5321 , news@e4ds.com




.jpg)

